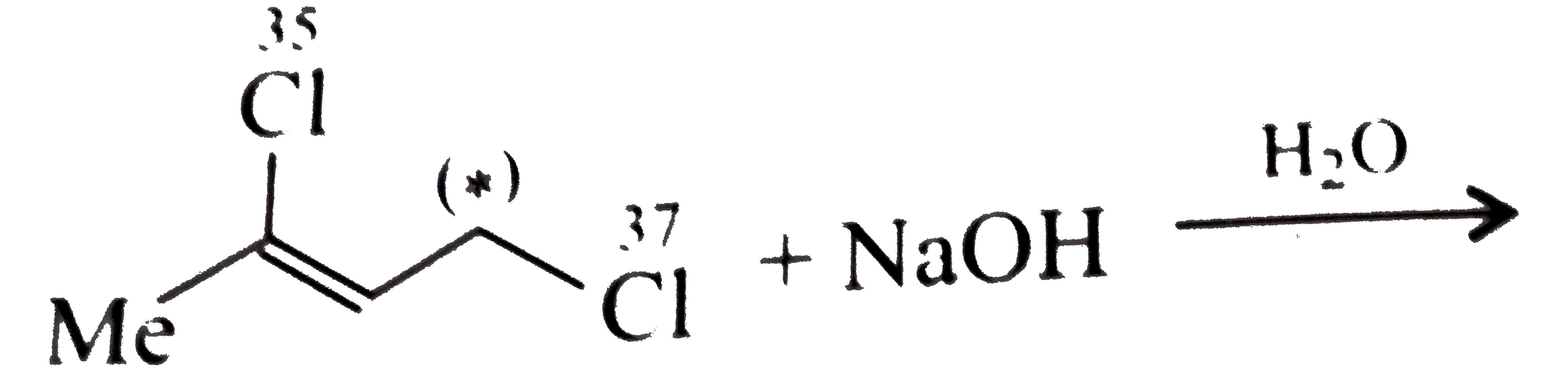
Will be equal to (1 atom x 65 grams/mole of zinc) + (two atoms x 14 grams/mole The brackets multiplied by the subscript two). Multiplied by the subscript two) and six atoms of oxygen (three atoms in Have one atom of zinc, two atoms of nitrogen (one atom inside the brackets (1 atom x 56 grams/mole Fe) + (2 atoms x 35.5 grams/mole of chlorine) = 127 grams/mole of iron (II) chloride For example, in iron (II) chloride, or FeCl 2, you have one atom of iron and two atoms of chlorine. If you have a subscript in a chemical formula, then you multiply the number of atoms of anything next to that subscript by the number of the subscript.įor most compounds, this is easy. (1 atom x 23 grams/mole Na) + (1 atom x 35.5 grams/mole Cl) = 58.5 grams/mole NaClĢ. If we write this as a calculation, it looks like this: If we have a chemical compound like NaCl, the molar mass will be equal to the molar mass of one atom of sodium plus the molar mass of one atom of chlorine. Molar masses of chemical compounds are equal to the sums of the molar masses of all the atoms in one molecule of that compound. To do this, we need to remember a few rules.ġ. How can I find the molar mass of a chemical compound?įor any chemical compound that's not an element, we need to find the molar mass from the chemical formula. Still, aside from the exceptions above, all elements have the same molar mass as the atomic masses on the periodic table. phosphorus is normally found in clumps of four atoms, P 4, and sulfur is found in clumps of eight atoms, or S 8. That each molecule of the element has two atoms of that element stuck together.Īs a result, the formula of hydrogen is H 2, nitrogen is N 2, etc. In the case of hydrogen, nitrogen, oxygen,įluorine, chlorine, bromine, and iodine, the element is diatomic, meaning In some cases, the element is usually found in a differentįorm than just one unbonded atom. So, in our example, carbon has a molar mass of 12.01 grams per mole. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. How can I find the molar mass of an element? 
It is also sometimes called: Molecular Mass, Molecular Weight, Formula Mass, or Formula Weight. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Chromium nitrate is used in the production of alkali metal-free catalysts and in pickling.Ĭhromium nitrate can be prepared by dissolving chromium oxide in nitric acid.Molar mass is the mass (in atomic mass units) of one mole of a of a substance. The red-violet hydrate is highly soluble in water. The anhydrous salt forms green crystals and is very soluble in water (in contrast to anhydrous chromium(III) chloride which dissolves very slowly except under special conditions). The chromium centers are bound to six aquo ligands, and the remaining volume of the solid is occupied by three nitrate anions and three molecules of water of crystallization. 3H 2O - betray a simple structure of this material.The relatively complicated formula - (NO 3) 3 It is common in academic laboratories for the synthesis of chromium coordination complexes. Chromium(III) nitrate compounds are of a limited commercial importance, finding some applications in the dyeing industry. Most common is the dark violet hygroscopic solid. Chromium(III) nitrate describes several inorganic compounds consisting of chromium, nitrate and varying amounts of water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
